
Yet if triumph in this scientific, corporate, and nationalistic contest is defined as the first vaccine to get approved and administered on a large scale, the victory is still anyone’s to win. The final phase will require navigating the maze of regulators, scattered across continents and agencies, who will determine when, which, and where shots of the most promising candidates will be approved, produced, and distributed. Indeed, Pfizer-BioNTech said it would file for the first approval of a Covid shot with the U.S. Food and Drug Administration.
These gatekeepers are an overlooked logistical reality of the vaccine contest. This isn’t just a single race among a handful of corporate front-runners but a more fractured process in which individual nations hold the power of approval. Yes, the FDA is important, but the contours of a splintered final push are already coming into focus—and they are global.
This story is from the November 23 - 30, 2020 edition of Bloomberg Businessweek.
Start your 7-day Magzter GOLD free trial to access thousands of curated premium stories, and 8,500+ magazines and newspapers.
Already a subscriber ? Sign In
This story is from the November 23 - 30, 2020 edition of Bloomberg Businessweek.
Start your 7-day Magzter GOLD free trial to access thousands of curated premium stories, and 8,500+ magazines and newspapers.
Already a subscriber? Sign In

Instagram's Founders Say It's Time for a New Social App
The rise of AI and the fall of Twitter could create opportunities for upstarts

Running in Circles
A subscription running shoe program aims to fight footwear waste

What I Learned Working at a Hawaiien Mega-Resort
Nine wild secrets from the staff at Turtle Bay, who have to manage everyone from haughty honeymooners to go-go-dancing golfers.

How Noma Will Blossom In Kyoto
The best restaurant in the world just began its second pop-up in Japan. Here's what's cooking

The Last-Mover Problem
A startup called Sennder is trying to bring an extremely tech-resistant industry into the age of apps

Tick Tock, TikTok
The US thinks the Chinese-owned social media app is a major national security risk. TikTok is running out of ways to avoid a ban
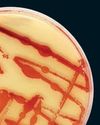
Cleaner Clothing Dye, Made From Bacteria
A UK company produces colors with less water than conventional methods and no toxic chemicals

Pumping Heat in Hamburg
The German port city plans to store hot water underground and bring it up to heat homes in the winter

Sustainability: Calamari's Climate Edge
Squid's ability to flourish in warmer waters makes it fitting for a diet for the changing environment

New Money, New Problems
In Naples, an influx of wealthy is displacing out-of-towners lower-income workers